Bioengineering PhD alumni Nick Fawzi and Sapun Parekh started a conversation about something completely different and ended up with a grant from the Human Frontier Science Program to study membraneless organelles.
Nicolas Fawzi is an Assistant Professor at Brown University, who received his Bioengineering PhD in 2007 working in Professor Teresa Head-Gordon’s lab. Sapun Parekh is a Group Leader at Max Planck Institute for Polymer Research in Mainz, Germany, who received his BioE PhD in 2008 working just upstairs in Professor Dan Fletcher’s lab.
“We never would have done this if we hadn’t met as PhD students in the same entering class together at Berkeley and in the Berkeley-UCSF program,” said Fawzi.
Their joint project is “Structure and biophysics of disordered domains mediating RNP granules: from atoms to cells”, funded with a $750,000 grant by the Human Frontier Science Program, a private foundation funding global collaborations. HFSP research grants provide 3 years of support for international teams involving at least two countries. “The project started when Nick saw my work on lipid (fatty) droplets in cells and initially read the paper title as liquid droplets, which he has been working on. Fortunately, we started talking and realized that the tools I used to see these droplets in cells would provide a unique slant on a good problem,” said Parekh.
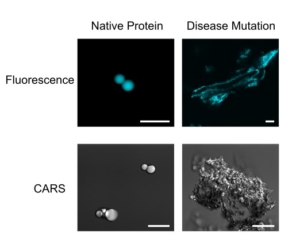
The team will create a novel molecular toolbox and experimental protocols to to observe, with residue-by-residue detail, the protein-protein interactions and structural changes caused by disease-causing missense mutations in human stress granules both in vitro and in cells.
Stress granules are only one example of a broad array of protein/RNA-containing “membraneless organelles”, assemblies long observed by microscopy inside cells but whose architecture remains poorly understood. Importantly, stress granule proteins are associated with the inclusions (solid clumps) observed in many neurodegenerative diseases including ALS (Lou Gehrig’s disease). By applying in-cell vibrational and FRET imaging combined with NMR spectroscopy to examine disease-associated variants, they will probe the underlying physico-chemical protein changes underlying disease-associated changes in granule properties. Their methods will be immediately applicable to other membraneless organelles and phase separated structures and their disease-associated changes.
Great, and unexpected, things happen when scientists talk!
